- HOME
- WHO WE ARE
- NEWS AND DATES
- GOODEIDS
- PHYLOGENY
- ARTIFICIAL KEY
- GOODEID SPECIES
- BIOLOGY
- ENVIRONMENT
- CONSERVATION
- PROFUNDULIDS
- MEMBERS AREA
Crenichthys nevadae
English Name:
Duckwater Railroad Valley Springfish
Original Description:
HUBBS, C. L. (1932): Studies of the fishes of the order Cyprinodontes. XII. A new genus related to Empetrichthys. Occasional Papers of the Museum of Zoology University of Michigan. Nr. 252: pp 1 - 5
Holotype:
Collection-number: Museum of Comparative Zoology - Harvard University, Cat. No. MCZ-32948.
The Holotype is an adult female, 44mm standard length, collected by the entomologist Charles Thomas Brues and his wife on July 21st, 1930. The only other fish taken with the Holotype was a female Paratype (UMMZ-95024) of 29mm SL, deposited in the University of Michigan Museum and Zoology.
The left picture shows the female Holotype of this species with a drawing of teeth, the right one a painting of a male by Joseph R. Tomelleri:
Terra typica:
The types were collected in an isolated warm spring (probably Big Warm Spring) at Duckwater, Nye County, Nevada, near the north end of Warm Spring Valley, 25.7km west and 74km south of Ely. Following the collectors, the species was abundant. These springs are located in the Duckwater Reservation of the native people of Tsaidüka meaning "Eaters of Tule (Schoenoplectus acutus) in Shoshone.
Etymology:
This species' name nevadae refers to the state of Nevada (USA) where this species is endemic.
The genus was erected by Hubbs in 1932 with the generic name referring to the typical habitat of this genus, namely springs. It is derived from the ancient Greek with κρήνη (kréne or créne) meaning spring and ἰχθύς (íchthús, íchthýs), the Greek word for fish. The name of the genus simply means "Springfish".
Synonyms:
none
Distribution and ESU's:
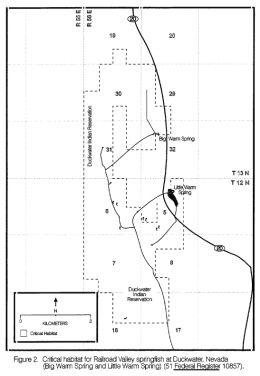
The former Crenichthys nevadae was historically known from two isolated spring areas located in the range of the ancient Lake Railroad and being separated from each other by approximately 43km beeline. Following last phylogenetic results (Campbell and Piller, 2017), the southern population will have to be described as a separate species, so we focus here only on the northern population, the "true" Crenichthys nevadae. This northern area is situated on the Duckwater Shoshone Indian Reservation containing two springs: Big Warm Spring and Little Warm Spring. The population of Big Warm Spring was extirpated in the late 1990' s and has been reestablished in 2007.
ESU ist short for Evolutionarily Significant Unit. Each unit expresses an isolated population with different genetic characteristics within one species. ESU's can be defined by Molecular genetics, Morphology and/or Zoogeography and help in indicating different phylogenetic lineages within a species. The abbreviation for an ESU is composed of three letters of the genus, followed by the first two letters of the species name and an ongoing number in each species.
In the subfamily Empetrichyinae, no ESU's were in use so far. To align with the livebearing subfamily Goodeinae, the GWG wants to bring a system in use based on John Lyons' naming for this subfamily. In Crenichthys nevadae, we actually (until the new species is described) distinguish two ESU's based on genetics. Crene1 encompasses the spring populations of Duckwater, so the "true" C. nevadae, while Crene2 is in use for the populations of the springs near Locke's Ranch and nearby refuges, that belong to a separate species (Crenichthys cf. nevadae).
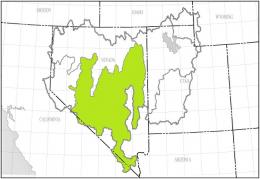
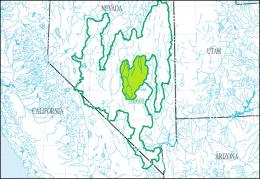
The left map shows the Central Nevada Desert Basins Accounting Unit (HUC 160600) of the Great Basin Region (HUC 16), the right map the Hot Creek-Railroad Valleys Cataloging Unit (HUC 16060012):
Status :
International Union for Conservation of Nature (IUCN): Vulnerable
U.S. Fish & Wildlife Service (ECOS): Threatened
Habitat:
The Railroad Valley springfish is found in warm spring pools, outflow streams, and adjacent marshes. Outflows tend to be shallow and less than 1m wide except for Big Warm Spring, which is several yards wide. The substrate of the head pools is typically sand, gravel or pebble with some decaying organic matter. Portions of some outflows contain dense mats of a nitrogen-fixing blue-green algae. Dense vegetation lines the pools and out-flows; salt grass is common in marshy areas.
For more specific information, we took the habitat descriptions from the Railroad Valley Springfish Recovery Plan (1997), slightly modified: Big Warm Spring is the largest spring in Railroad Valley with a discharge that varied from 23.8m³p.m. in 1912 to 22.1m³p.m. in 1963 (Garside & Schilling, 1979). Its main spring pool is approximately 24m in diameter (Williams, 1986). Water temperature at the spring pool has varied between 30 and 33°C. The outflow from Big Warm Spring is immediately divided into two channels by a concrete diversion structure. The south outflow extends approximately 1,300m before cascading over a bluff and joining Duckwater Creek. The north outflow extends approximately 350m before entering an underground pipe. Little Warm Spring, located approximately 1.6km south of Big Warm Spring, discharged 1.1m³p.m. in 1966 (Garside & Schilling, 1979). Water temperatures at the source were relatively constant at 33°C. The outflow from Little Warm Spring historically was highly vegetated with undercut banks, and flowed into a 7.5ha marsh. In 1984, a ditch was dug along the eastern side of the marsh to drain it in preparation for agriculture (Williams, 1986). This ditch was still in use, although no agricultural development had occurred at the marsh. A second, smaller ditch carried water to the west where it flew over a small bluff approximately 800m from its source.
Biology:
Breeding occurs year-round but ovary production documented from Big Warm Spring was greatest during the summer, declined in spring and fall, and was poorly developed in winter. When water temperatures were especially high, no larval fish were produced in any season. Reproduction seems to be severely restricted in water temperatures above 35°C. The ratio of males to females is even in the spring but the number of females almost doubles in the summer and fall.
Diet:
Primarily feeding herbivorous in spring, observations in summer show that Crenichthys cf. nevadae from Big Spring at Locke's Ranch is then carnivorous, preferring ostracods (Williams & Williams, 1989), but regarding the feeding habits of closely related inhabitants of thermal springs, Crenichthys nevadae probably is an opportunistic omnivore, with animal food representing around 2/3 or more of its consumption during the summer, primarily consisting of gastropods. Plant consumption is mostly filamentous algae. While the intestine length is consistent with an omnivore, Sigler & Sigler suggest that the high water temperatures of the Springfish habitat may demand the higher energy available with animal food.
Size:
The maximum known TL is 72mm (Williams, 1986).
Colouration:
The colouration of Crenichthys nevadae is quite similar to those of the other species of the genus, especially to Crenichthys cf. nevadae with which is shares a single lateral band of black blotches instead of two like in Crenichthys baileyi and its sister species from the Muddy River. The colour of the back is brownish grey to olive with the flanks partly in yellow to fully golden in adult males. The belly is white and a single lateral row blotches is present, forming a dark band in adults and almost disappears in older individuals. Fully grown up male specimens have a dark back, golden flanks and a white to creme belly, while the females' colour is paler. The operculum is golden and fins milky to dusky grey, confined by dark margins. The colouration is intensifying in excitement, a dark medial band on the back of male fish is shown, extending from the mouth to the dorsal fin origin and the golden colour gets striking.
Sexual Dimorphism:
The sexes differ mainly in shape, fins and colouration. Males are higher in body and appear stronger, anal and dorsal fins are longer and have a more sailfin shape while the these fins of females are shorter and the fins are rather shaped like a rhombus. Male unpair fins express a broad dark to black terminal band and are coloured more intensively while those of females stay mainly clear or are slightly coloured. Males have their lateral band broader than females but less distinct and the medial stripe in males is darker and more prominant, especially during courtship.
Remarks:
The following information is mostly taken from the Railroad Valley Springfish Recovery Plan (1997) and from the 5-year review from 2009, slightly modified and supplemented.
These small fish were a traditional food source for the Shoshones in this area prior to non-native settlement in the late 1800's. The population in the Big Warm Spring was abundant during the 1930’s, but became rare in the early 1980's, following the introduction of nonnative fishes and the installation of a catfish rearing facility (Ictalurus punctatus). The species persisted in Big Warm Spring in extremely limited numbers for over 10 years, then the population rebounded between 1994 and 1996, while the catfish facility was inoperative. Visual observations during April and sampling conducted during July 1996 suggested that Railroad Valley Springfish was the most abundant fish in the system during the late winter and spring, but by midsummer nonnative fishes were the most abundant ones, mainly Guppies, Sailfin Mollies, and Mosquitofish. During 1986-2000, Big Warm Spring and the associated outflows were modified annually to maintain and improve the catfish aquaculture facility and maintain flows for irrigation. In 2003, the Redbellied Tilapia (Tilapia zillii) was the dominant fish species in Big Warm Spring with catfish and Mollies being the other most common fish species (Nielsen, 2003), and probably causing the extinction of the Springfish in Big Warm Spring. Intensive minnowtrapping in 2003 revealed not a single Springfish within the spring pool, outflow channels, and at the catfish facility. Habitat restoration and nonnative fish eradication activities took place in 2005 and 2006 and the habitat was declared free of nonnative predatory fish (Mosquitofish and Mollies were still there) and again a suitable habitat for Crenichthys nevadae. Railroad Valley Springfish were restored to Big Warm Spring on September 26th, 2007. Stocking has been ongoing since then and, to date, 988 specimens have been relocated from Little Warm Spring, School Spring and Sugar Shack Spring into Big Warm Spring (Hobbs, 2007 and 2008). Mollies still occur on the Duckwater populations but an agreement with U.S. Fish and Wildlife Service and the Duckwater Tribe has hired some tribal technicians (high school kids) to trap and remove mollies to help keep the invasive population low. Other threats are over use of groundwater for oil and gas leases, and proposed fracking (Guadalupe, pers. com., 2021).
The number of fish in Little Warm Spring increased in 1934, and the Springfish remained “common” despite the draining of an associated marsh and diversion of the spring outflow into two ditches. In 1986, Railroad Valley Springfish occupied the spring pool and both ditches, although densities were greatest in the west ditch. In June 1989, very few Railroad Valley springfish were observed at Little Warm Spring, but the population rebounded by 1992. Data collected during August 1994 and August 1996 suggest the Railroad Valley springfish population fluctuated between these years, but remained abundant to common at Little Warm Spring. Restoration efforts in 2007 have redirected the outflow stream back to the historical channel and through a series of created wetlands in an attempt to restore the Little Warm Spring system to a more natural state. Surveys in 2008 found the species in channel and wetlands. Visual observations and surveys at Little Warm Spring indicate that the population's abundance is low and fluctuates from year to year, but has remained relatively stable since 2003 (Hobbs, 2007).
Three additional populations have been documented on the Reservation since the publication of the 1997 Recovery Plan. The best available information suggests that these are historical populations that were part of a once larger population found throughout the Duckwater Creek watershed when the natural historical flow patterns of many spring systems provided habitat connectivity. These populations are in close proximity to other historical sites on the Reservation, and the associated headwater springs are relatively isolated. One population is in several small unnamed Spring systems that flow from the Duckwater Falls bluff. The northern most spring along the bluff, School Spring, produces less than 0.04m³p.s. of water and flows from the bluff towards the west and then turns to the south where it flows under the main road. It then becomes highly channelized and is eventually diverted for flood irrigation to pasture land on the central part of the Reservation. The other two additional springs containing Springfish are located on Sugar Shack Road to the south of Duckwater Falls Road. One spring originates from the bluff to the east of Sugar Shack Road and the other originates to the west of the road. They are collectively known as the Sugar Shack Road Springs by locals; however, there is no official name for them on historic maps. Less than 0.03m³p.s. of water originates from the Sugar Shack Road Springs; therefore, traditional Springfish survey methods are virtually impossible due to shallow water depths. Visual observations of springfish distribution in 2005 indicated that the populations at the Sugar Shack Road Springs were present only in the spring pools (Hobbs, 2007). In 2005 and 2006, NDOW (Hobbs, 2007) visually estimated the population in School Spring to be 100-300 fish. Visual estimates in 2005 of the two populations of fish at the Sugar Shack Road Springs ranged from 50 to 200 fish in each spring. Subsequent visual surveys indicate that all three of these recently identified locations continue to support populations of Springfish (Morrell et al., 2007).
In 1947, Nevada State Fish and Game Commission (now called Nevada Department of Wildlife) personnel (Thomas Trelease) released six Railroad Valley Springfish from Big Warm Spring into ponds on a private property at Sodaville, Mineral County. The Springfish occupied the southernmost of the two spring groups at Sodaville. In 1991, this population was estimated to contain 250 fish, based on visual observations (Sevon, 1991). Mark-and-recapture trapping efforts conducted in July 1994, June 1995, and June 1996 resulted in population estimates of 125, 81, and 27 individuals, respectively (Elliott, 1994, 1995; Crawforth & Drake, 1996). This population was declining as open water habitat became overgrown with emergent aquatic vegetation. No fish surveys of the Sodaville Springs have been conducted since 2002. This spring system is owned by a private landowner who operated an Australian lobster (Metanephrops australiensis) aquaculture facility downstream of the spring pools. Springfish populations were significantly reduced within the Spring pools and outflow channels because of habitat modification, but some fish were able to access and survive in the facility raceways. Springfish were observed in the raceways during 2002 (B. Nielsen, 2002). In 2004, the Nevada Department of Wildlife (NDOW) revoked the landowner’s State aquaculture permit and assisted the landowner with decommissioning the facility by removing a portion of the raceways and pipelines and eradicating the lobster. The fish have likely been extirpated at Sodaville Springs because of the removal of the raceways and the lack of suitable habitat in the spring pools. No fish were found until 2020, so the Sodaville population definitely went extinct (Guadalupe, pers. com., 2021).
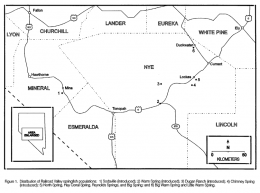
The left picture shows the natural habitats and known introductions of Crenichthys nevadae and its sister species with numbers 6 (native range) and 1 (Sodaville introduction site) belonging to Crenichthys nevadae, the right picture a more detailed map of Duckwater showing Big and Little Warm Springs:
Species of the subfamily Empetrichthyinae are oviparous fishes and share the lack of pelvic fins, while the subfamily Goodeinae is livebearing. Lynne Parenti (1981) finally proposed this subfamily as sister group to the Goodeinae and encompassed both in the family Goodeidae. Meanwhile the monophyly of the family Goodeidae and the narrow relationship between both subfamilies has been supported through several studies since the 1980's and is not doubted any longer.
In 2017, D. Cooper Campbell and Kyle R. Piller pubished a phylogenetic study of the Empetrichthyinae. Concerning Crenichthys nevadae, individuals from seven localities were included, namely the four springs on Lockes Ranch, Hot Terracy Spring, and from the Duckwater Reservation School Spring and Little Warm Spring. The studies were separatly done on the mitochondrial gene Cytochrome b and three nucleotide sequences, namely S7 intron-1, S8 intron-4, and P0 intron-1. Concerning cytb, the authors found two clades corresponding with the two distribution areas and genetic distances between 1.6 and 1.8%. Concerning the three introns, a clade formed by the southern ones from Lockes Ranch and Hot Terrace Spring was recognized, and Haplotype network analysis recognized two separate clades as well. Phylogeny from species tree analysis based on cytb hypothesis following Bayesian statistics supports a model of two species. Taking in consideration genetic distances in the cytb gene that separate species in the Goodeinae and the result of the tree analyis, we follow a conservative approach here and recognize two species: Crenichthys nevadae from the type locality (Big Warm Spring, Duckwater) and Crenichthys cf. nevadae from the southern habitats next to Lockes Ranch.
Husbandry:
Looking on the biotopes of Crenichthys nevadae, they suggest the species may prefer a habitat with moderate to swift current, structured with gravel, rocks, roots, branches, fallen leaves and river bank vegetation. Fry is rarely eaten, but it may depend on the quantity and quality of food and on the number of places to hide. When several different stages of juveniles occur, fry will be neglected, so it makes eventually sense to add separate brought up fry to the group with a size of 1.5 or 2cm to provide these stages and get a flock breeding colony.
The recommended tank size is at least 100 liters, bigger tanks with a generous base and little height (25cm are enough) are better for sure. With rocks and vegetation in the corners and backside of the tank well structured tanks combined with some roots and/or wood seem to do best with this species. The current should be moderate or swift, but the species can deal wih low oxygen values (down to 3mg/l).
In the wild, the species feeds mainly from small or middle-sized invertebrates like bloodworms or insect larvae, gammarids, ostracods, but also algae, aufwuchs, detritus and other organic matters, so feeding with similar food, water fleas, Mysids and other food from animalistic sources as well as stones and pebbles covered with green algae will be best for this omnivorous fish. In aquarium, it feeds very well from flake food, granulate and tablets, additionally given Nauplia of Brine Shrimps are eaten greedy. Additionally, as aufwuchs and green algae are taken in the natural habitat, providing some kind of vegetables like boiled peas is a good food supplement. The species is not shy.
Concerning water quality, this species is in need of bigger water changes (60-80% every week) like most of the Goodeids, especially river and spring inhabiting species, so an automatic water changing system can be helpful, but in contrast to most of the Goodeid species from Mexico, it needs to be temperated and the oxygen level could be low. This species does best at temperatures between 25 and 29°C though it is able to withstand lower temperatures down to even 12°C for a short period.
This species is doing very well when is kept in the open from spring to fall, starting when the water temperature by night does no more deceed 18°C and colder periods are no longer expected. Bring them out in the early afternoon, the time of the day with the highest water temperature. Bring the fish in before the water temperature deceeds 18°C by night and keep them at this temperature for the first days, then slowly raise the temperature to the convenient range of this species over the winter time.
Populations in holding:
Here each species are assigned populations of fish in husbandry and in brackets aliases of these locations to assist in identifying own stocks. Each population is assigned a unique Population-ID, composed by the ESU, the subbasin where this population is occurring (three capital letters) and a unique location identifier.
Populations in holding:
none